1. The bubble model
Models of crystal structure have been described from time to time in which the atoms are represented by small floating or suspended magnets, or by circular disks floating on a water surface and held together by the forces of capillary attraction. These models have certain disadvantages; for instance, in the case of floating objects in contact, frictional forces impede their free relative movement. A more serious disadvantage is that the number of components is limited, for a large number of components is required in order to approach the state of affairs in a real crystal. The present paper describes the behaviour of a model in which the atoms are represented by small bubbles from 2.0 to 0.1 mm. in diameter floating on the surface of a soap solution. These small bubbles are sufficiently persistent for experiments lasting an hour or more, they slide past each other without friction, and they can be produced in large numbers. Some of the illustrations in this paper were taken from assemblages of bubbles numbering 100,000 or more. The model most nearly represents the behaviour of a metal structure, because the bubbles are of one type only and are held together by a general capillary attraction, which represents the binding force of the free electrons in the metal. A brief description of the model has been given in the Journal of Scientific Instruments (Bragg 1942b).
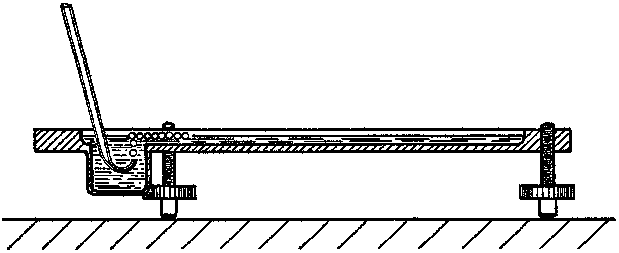
figure 1. Apparatus for producing rafts of bubbles.
Более 800 000 книг и аудиокниг! 📚
Получи 2 месяца Литрес Подписки в подарок и наслаждайся неограниченным чтением
ПОЛУЧИТЬ ПОДАРОК